42+ Geiger and marsden experiment animation ideas in 2021
Home » Background » 42+ Geiger and marsden experiment animation ideas in 2021Your Geiger and marsden experiment animation images are ready in this website. Geiger and marsden experiment animation are a topic that is being searched for and liked by netizens now. You can Get the Geiger and marsden experiment animation files here. Download all royalty-free images.
If you’re looking for geiger and marsden experiment animation images information related to the geiger and marsden experiment animation keyword, you have come to the right site. Our website always provides you with suggestions for seeking the maximum quality video and image content, please kindly search and find more informative video articles and graphics that fit your interests.
Geiger And Marsden Experiment Animation. What is mass defect. They deduced this by measuring how an. Rutherford suggested that Marsden look for alpha-particle scattering at large angles. It was carried out by his assistants Hans Geiger and Ernest Marsden.
 Rutherford S Gold Foil Experiment From sciencefacts.net
Rutherford S Gold Foil Experiment From sciencefacts.net
They deduced this by measuring how an alpha particlealpha particle. If the light of wavelength 6 8 0 0 A is incident on the surface of metal find threshold frequency and incident frequency. The GeigerMarsden experiments also called the Rutherford gold foil experiment were a landmark series of experiments by which scientists discovered that every atom has a nucleus where all of its positive charge and most of its mass is concentrated. John Dalton published his ideas about atoms. The GeigerMarsden experiments were a landmark series of experiments by which scientists discovered that every atom has a nucleus where all of its positive charge and most of its mass is concentrated. The Rutherford Experiment.
He didnt think it very likely.
John Dalton published his ideas about atoms. Pradeep Kshetrapal Tutorials Point India Private Limited. This classic diffraction experiment was conducted in 1911 by Hans Geiger and Ernest Marsden at the suggestion of Ernest Rutherford. How did Rutherford figure out the structure of the atom without being able to see it. The GeigerMarsden experiments also called the Rutherford gold foil experiment were a landmark series of experiments by which scientists discovered that every atom has a nucleus where all of its positive charge and most of its mass is concentrated. If the light of wavelength 6 8 0 0 A is incident on the surface of metal find threshold frequency and incident frequency.
 Source: pngegg.com
Source: pngegg.com
The Rutherford Experiment helps us to understand the structure of atoms. This video tutorial has been designed to suit the curriculum of CBSE Class 12 students. 506 1910 621 the scattering was found to vary at a rate more nearly proportional to the inverse fourth power of the velocity than to any other integral power. In 1909 Geiger needed an experiment for a research student Ernest Marsden. He thought that all matter is made of atoms which he imagined as tiny solid balls.
 Source: youtube.com
Source: youtube.com
This video tutorial has been designed to suit the curriculum of CBSE Class 12 students. Will there be an emission of. In the experiment Rutherford sent a beam of alpha particles helium nuclei emitted from a radioactive source against a thin gold foil the thickness of about 00004 mm corresponding to about 1000 atoms. Simulate the famous experiment in which he disproved the Plum Pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. How did Rutherford figure out the structure of the atom without being able to see it.
 Source: pinterest.com
Source: pinterest.com
They deduced this after measuring how an alpha particle beam is scattered when it strikes a thin metal foil. Pradeep Kshetrapal Tutorials Point India Private Limited. In this video tutorial we will discuss topics such as the contents of atom Geiger and Marsden Experiment Primary shape of atom impact parameter etc. It was carried out by his assistants Hans Geiger and Ernest Marsden. They deduced this by measuring how an alpha particlealpha particle.
 Source: pinterest.com
Source: pinterest.com
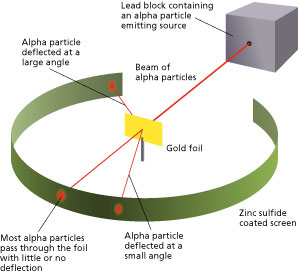
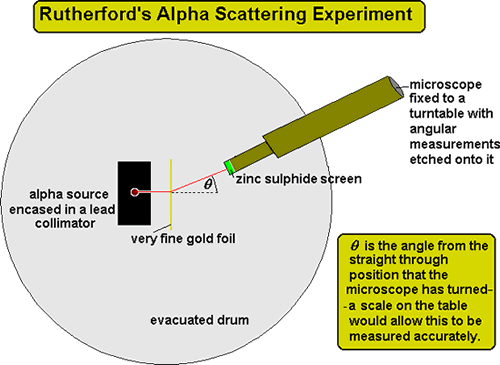
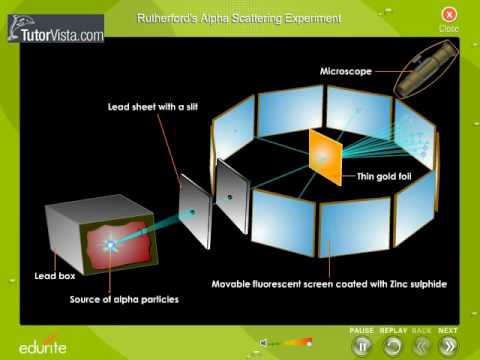
He thought that all matter is made of atoms which he imagined as tiny solid balls. The experiments were performed between 1908 and 1913 by Hans Geiger and Ernest Marsden under the direction of Ernest Rutherford at the Physical Laboratories of the University of Manchester. The Rutherford Experiment helps us to understand the structure of atoms. With the help of a neat labelled diagram describe the Geiger- Marsden experiment. In this experiment a narrow beam of α-particles from a radioactive source was incident on a gold foil.

The experiment showed the presence of a positively charged kernel. With the help of a neat labelled diagram describe the Geiger- Marsden experiment. Simulate the famous experiment in which he disproved the Plum Pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. The photoelectric work function for a metal surface is 23 eV. The scattered α-particles were detected by the detector fixed on a rotating stand.
 Source: physicsopenlab.org
Source: physicsopenlab.org
The GeigerMarsden experiments were a landmark series of experiments by which scientists discovered that every atom has a nucleus where all of its positive charge and most of its mass is concentrated. Details about the experiment and how to operate the tutorial are provided beneath the applet window. This video tutorial has been designed to suit the curriculum of CBSE Class 12 students. What is mass defect. It was carried out by his assistants Hans Geiger and Ernest Marsden.
 Source: slideplayer.com
Source: slideplayer.com
The GeigerMarsden experiments were a landmark series of experiments by which scientists discovered that every atom has a nucleus where all of its positive charge and most of its mass is concentrated. This video tutorial has been designed to suit the curriculum of CBSE Class 12 students. They deduced this by measuring how an. Will there be an emission of. Rutherfords scattering experiment A scientist called Ernest Rutherford designed an experiment to test the plum pudding model.
 Source: pinterest.com
Source: pinterest.com
Rutherford suggested that Marsden look for alpha-particle scattering at large angles. How did Rutherford figure out the structure of the atom without being able to see it. Several experiments were made and in every case H. It was carried out by his assistants Hans Geiger and Ernest Marsden. Details about the experiment and how to operate the tutorial are provided beneath the applet window.

What is mass defect. This classic diffraction experiment was conducted in 1911 by Hans Geiger and Ernest Marsden at the suggestion of Ernest Rutherford. Simulate the famous experiment in which he disproved the Plum Pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. The GeigerMarsden experiments also called the Rutherford gold foil experiment were a landmark series of experiments by which scientists discovered that every atom contains a nucleus where its positive charge and most of its mass are concentrated. The Rutherford Experiment.
 Source: sciencefacts.net
Source: sciencefacts.net
506 1910 621 the scattering was found to vary at a rate more nearly proportional to the inverse fourth power of the velocity than to any other integral power. The GeigerMarsden experiments also called the Rutherford gold foil experiment were a landmark series of experiments by which scientists discovered that every atom contains a nucleus where its positive charge and most of its mass are concentrated. What is mass defect. He didnt think it very likely. Several experiments were made and in every case H.
 Source: youtube.com
Source: youtube.com
Details about the experiment and how to operate the tutorial are provided beneath the applet window. They deduced this after measuring how an alpha particle beam is scattered when it strikes a thin metal foil. The GeigerMarsden experiments were a landmark series of experiments by which scientists discovered that every atom has a nucleus where all of its positive charge and most of its mass is concentrated. With the help of a neat labelled diagram describe the Geiger- Marsden experiment. The detector used had a zinc sulphide screen and a microscope.
 Source: favpng.com
Source: favpng.com
Will there be an emission of. Will there be an emission of. In this experiment a narrow beam of α-particles from a radioactive source was incident on a gold foil. What is mass defect. The GeigerMarsden experiments also called the Rutherford gold foil experiment were a landmark series of experiments by which scientists discovered that every atom has a nucleus where all of its positive charge and most of its mass is concentrated.
 Source: cyberphysics.co.uk
Source: cyberphysics.co.uk
The scattered α-particles were detected by the detector fixed on a rotating stand. The experiment showed the presence of a positively charged kernel. It was carried out by his assistants Hans Geiger and Ernest Marsden. The GeigerMarsden experiments also called the Rutherford gold foil experiment were a landmark series of experiments by which scientists discovered that every atom has a nucleus where all of its positive charge and most of its mass is concentrated. In 1909 Geiger needed an experiment for a research student Ernest Marsden.
 Source: youtube.com
Source: youtube.com
The GeigerMarsden experiments were a landmark series of experiments by which scientists discovered that every atom has a nucleus where all of its positive charge and most of its mass is concentrated. This classic diffraction experiment was conducted in 1911 by Hans Geiger and Ernest Marsden at the suggestion of Ernest Rutherford. Geiger-Marsden experiment Daltons model. He didnt think it very likely. Details about the experiment and how to operate the tutorial are provided beneath the applet window.
 Source: youtube.com
Source: youtube.com
Geiger-Marsden experiment Daltons model. The experiments were performed between 1908 and 1913 by Hans Geiger and Ernest Marsden under the direction of Ernest Rutherford at the Physical Laboratories of the University of Manchester. The GeigerMarsden experiments also called the Rutherford gold foil experiment were a landmark series of experiments by which scientists discovered that every atom contains a nucleus where its positive charge and most of its mass are concentrated. John Dalton published his ideas about atoms. In this experiment a narrow beam of α-particles from a radioactive source was incident on a gold foil.
 Source: sciencefacts.net
Source: sciencefacts.net
These classes will be helpful in preparing the students for their board examinations as well. They deduced this after measuring how an alpha particle beam is scattered when it strikes a thin metal foil. John Dalton published his ideas about atoms. The GeigerMarsden experiments were a landmark series of experiments by which scientists discovered that every atom has a nucleus where all of its positive charge and most of its mass is concentrated. What is mass defect.
 Source: physicsopenlab.org
Source: physicsopenlab.org
They deduced this by measuring how an alpha particlealpha particle. The GeigerMarsden experiments also called the Rutherford gold foil experiment were a landmark series of experiments by which scientists discovered that every atom has a nucleus where all of its positive charge and most of its mass is concentrated. The GeigerMarsden experiments also called the Rutherford gold foil experiment were a landmark series of experiments by which scientists discovered that every atom contains a nucleus where its positive charge and most of its mass are concentrated. The scattered α-particles were detected by the detector fixed on a rotating stand. This video tutorial has been designed to suit the curriculum of CBSE Class 12 students.
 Source: pinterest.com
Source: pinterest.com
It was carried out by his assistants Hans Geiger and Ernest Marsden. Simulate the famous experiment in which he disproved the Plum Pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. They deduced this by measuring how an alpha particle beam is scattered when it strikes a thin metal foil. The detector used had a zinc sulphide screen and a microscope. In the experiment Rutherford sent a beam of alpha particles helium nuclei emitted from a radioactive source against a thin gold foil the thickness of about 00004 mm corresponding to about 1000 atoms.
This site is an open community for users to submit their favorite wallpapers on the internet, all images or pictures in this website are for personal wallpaper use only, it is stricly prohibited to use this wallpaper for commercial purposes, if you are the author and find this image is shared without your permission, please kindly raise a DMCA report to Us.
If you find this site adventageous, please support us by sharing this posts to your own social media accounts like Facebook, Instagram and so on or you can also bookmark this blog page with the title geiger and marsden experiment animation by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.
Category
Related By Category
- 49+ Corn husk animals information
- 29++ Chinese year 1968 animal info
- 32++ Do animated gifs work in outlook info
- 22++ Animals in space nasa ideas
- 30+ Dragon nest animation information
- 20++ How to animate over live action ideas
- 20++ Chanel logo animation ideas
- 44++ Animals that went extinct in 2013 ideas
- 37++ Disney pixar animals information
- 31+ Bodyweight animal exercises info